With the entire world has come into a standstill because of the global pandemic novel coronavirus, everyone has been staying at home to fight against this virus. While the reports have claimed that about the vaccines which will come soon, researchers at Northwestern University have claimed that they have developed the world’s first wearable piece of technology that can monitor the symptoms of COVID-19.
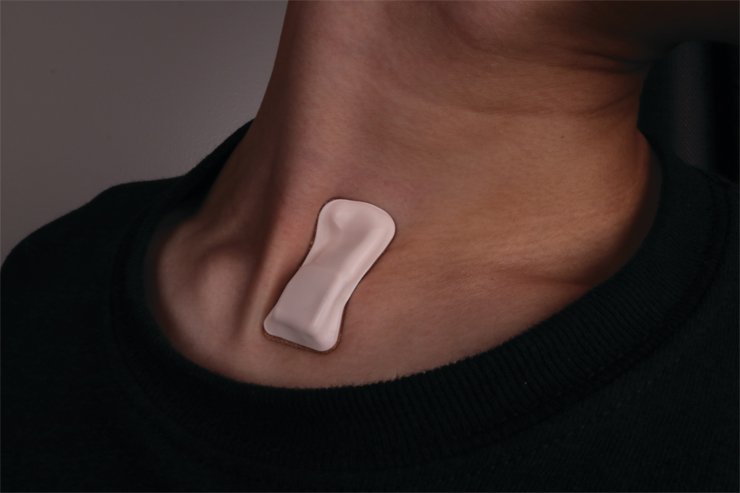
The wearable device which developed was a small, wearable patch that sticks on one’s throat 24/7. The device can measure symptoms like coughing and breathing patterns, heart rate, and body temperature.

The sensor will collect the data and deliver to the cloud server and it will be analyzed using an algorithm. The researchers also said that the algorithm is being updated on a constant basis to help doctors to track the progression of the disease as well.
The stamp-sized device can be used with ease
However, wearing a stamp-sized tech is certainly not necessary for everyone as of now. Moreover, this device isn’t available for commercial use yet too.
Moreover, the team behind the patch further said that many patients with Coronavirus witness their symptoms fully cured before it starts to get worse soon. They shared continuous monitoring can help physicians check in the early stages and help stop the virus from spreading.

In the near future, we can see frontline workers also wearing such a patch if the situation doesn’t get better any time soon. A device like this can be really helpful to them since they are at the risk of getting affected on a daily basis. As per the researchers, the patch can also be sterilized and reused too, so it’s also very cost-effective on the other hand.
As of now, more than 25 people have been using the patch for nearly about two weeks and the sensors have also pretty well managed to collect more than one terabyte of data. People who have been using the patch have already claimed that it’s very small and they don’t even notice it too.
However, it’ll be interesting to see how this product helps people and do well in the market where health officials and medical agencies can have the access to it once it’s fully ready to be used.
Source: Northwestern University
Follow Wat-Not on Facebook, Twitter, and Instagram